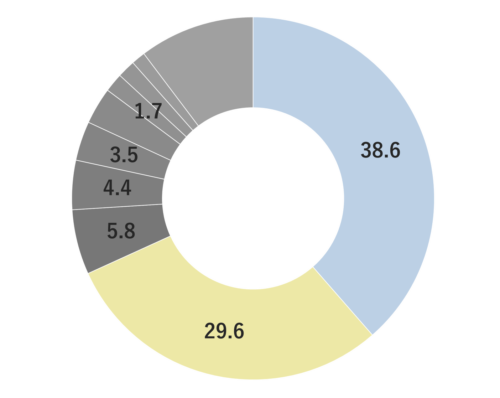
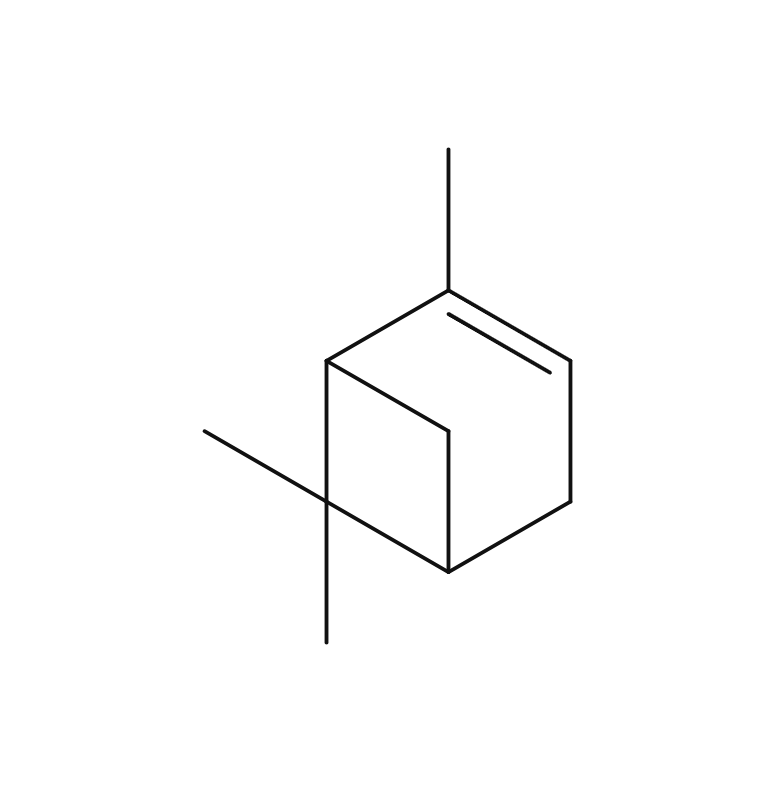
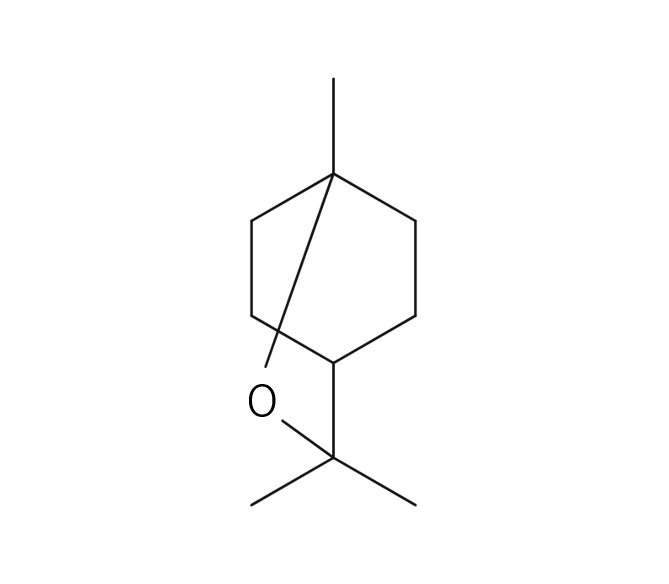
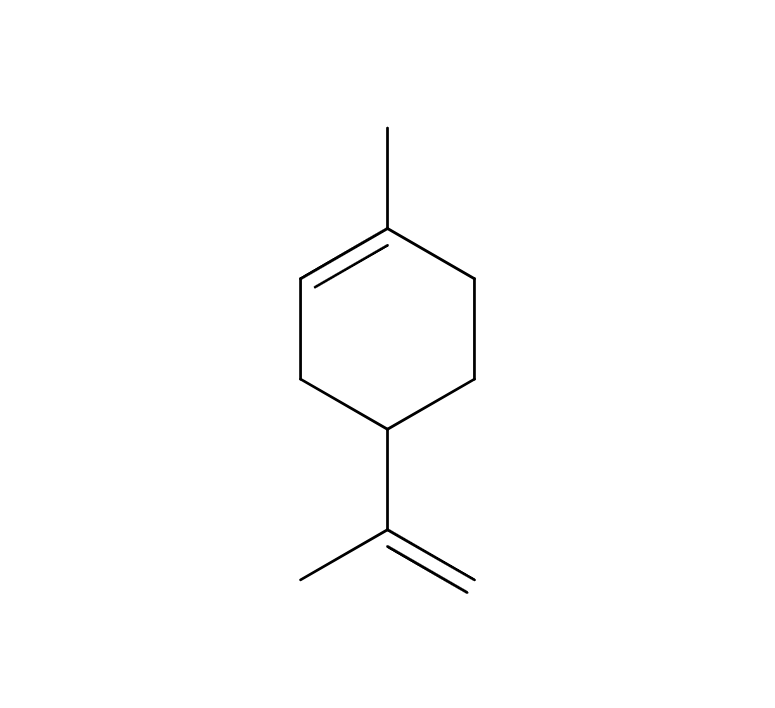
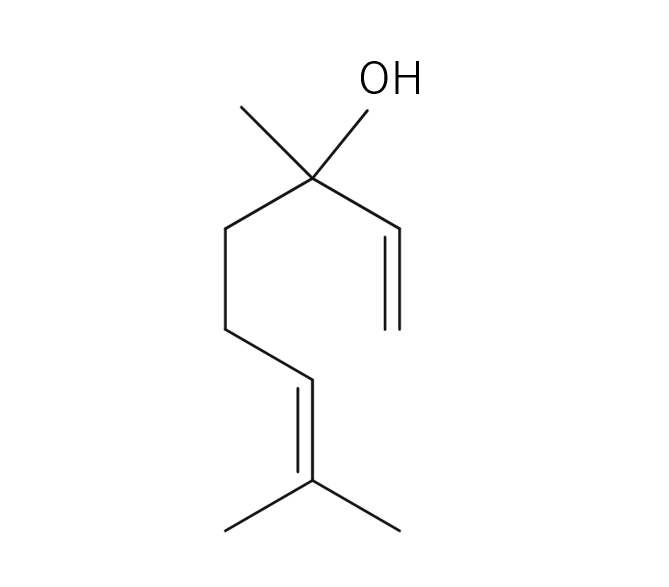
Cardamom is a perennial herb of the ginger family, growing up to 60 cm high. Its leaves are large, green, and oval-shaped, arranged alternately. The flowers can be either white or purple, and the three-chambered pods contain seeds. These seeds, known as cardamom, are used as spice and flavoring. The genus name “Elettaria” is believed to have originated from the local name of the Malabar region in Sri Lanka. Its type is known to be close to Ginger, Ply and Turmeric but the root lacks aroma; instead, the fragrance emanates from the small seeds and their pods. Cardamom boasts a rich history as a spice, having been introduced to Europe from India around the 2nd century BC.